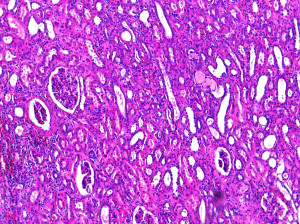
Kidney Cancer Samples
Table of Contents
Introduction | Classification | Diagnosis and Treatment | Considerations for Researchers seeking kidney cancer samples | References
Looking for human kidney cancer samples for use in your research? Contact us for a consultation.
Introduction
Kidney cancer is one of the ten most common cancers in the United States for both men and women, accounting in 2014 for an estimated 63,920 new cases (39,140 men and 24,780 women) and an estimated 13,860 deaths (8,900 men and 4,960 women), representing 3.8% of all new cancer cases and 2.4% of all cancer deaths (American Cancer Society 2011). Additionally, about 1.6% of individuals or 1 in 63 will develop kidney and renal pelvis cancer sometime during their lives (Ibid.). Regarding prevalence, it was also estimated that in 2011 there were 358,603 individuals living with kidney and renal pelvis cancer in the U.S (SEER 2005). New kidney and renal pelvis cancer rates have been steadily rising for the past ten years with an average of 1.6% per year, placing it in 2014 as the 8th most common cancer (SEER 2005). However, the rate seems to have leveled off over the last few years, which can probably be partly attributed to newer imaging tests such as CT scans and their ability to reveal cancer that may have remained hidden otherwise. During approximately the same ten year time frame, the death rates fell gradually by about 0.6% each year. Most individuals diagnosed are ages 55-64, with 64 being the median age at diagnosis. Kidney cancer is also more common among men than women (SEER 2005). About 64% of cases are localized, for which the survival rate is about 92%; 17% are regional (the cancer has spread to regional lymph nodes), for which the survival rate is about 65%; 16% are distant (the cancer has metastasized), for which the survival rate is about 12%; and the remainder of cases (3%) are unstaged (SEER 2005).
Lifestyle-related factors and kidney cancer risk:
- Smoking – carcinogens enter the bloodstream through the lungs, and the blood is filtered by the kidneys
- Obesity – may cause kidney cancer by causing hormonal changes
- Hypertension – mechanism unknown
- Family history – the relative risk for a sibling of an RCC patient is 2.5; hereditary syndromes are found in some cases (see above)
- Sex – RCC is roughly twice as common in men as in women, possibly due to an increased prevalence of smoking among men than woman and greater exposure to workplace chemicals among men than women. Another possibility is related to higher levels of androgens in men than in women. A recent study found that androgen receptor (AR) “enhanced the human kidney epithelial cell tumorigencity,” and “increased the proliferation rate of RCC cell lines,” (He, et al. 2014).
- Race – African Americans and Native Americans/Alaska Natives have slightly higher rates of RCC than whites, for unknown reasons (Urology Health, 2011).
- Advanced kidney disease – especially if the patient is on dialysis
- Workplace exposures – chemicals such as trichloroethylene and cadmium may increase the risk of developing kidney cancer
- Dietary intake of fruits and vegetables – there is an inverse correlation between development of kidney cancer and intake of fruits and vegetables
- Low cholesterol – has been linked with lower survival rates in kidney cancer patients
From: Wiley (2014).
Genetics of Kidney Cancer
Currently, only about 5-8% of kidney cancers are thought to have had an inherited basis. However, a recent Icelandic study found that nearly 60% of all cases of kidney cancer were in families that had two or more affected members, indicating that germline mutations may be much more commonly involved in what have been considered to be sporadic (non-inherited) cases than has previously been thought (Linehan, 2012).
Kidney cancer can be thought of as a metabolic disease because the kidney cancer genes VHL, MET, FLCN, FH, SDH, TSC1, TSC2, and TFE3 all affect the cell’s ability to sense oxygen, iron, nutrients, the Krebs cycle enzymes FH and SDH, and energy (Linehan, 2012).
Four major inherited autosomal dominant RCC syndromes have been identified (From: Linehan, 2012):
- von-Hippel-Lindau syndrome (VHL) – caused by an inherited mutation to the VHL tumor-suppressor gene.
- Hereditary leiomyomatosis and renal cell cancer (HLRCC) – caused by germline mutation in the FH gene. Individuals with HLRCC are at risk of developing type 2 papillary kidney cancer.
- Hereditary papillary renal cancer (HPRC) – caused by germline mutation of the MET gene on chromosome 7. Individuals with HPRC are at risk of bilateral, type 1 papillary kidney cancer.
- Birt-Hogg-Dubé syndrome (BHD) – associated with mutations in the FLCN gene.
Gene mutations of kidney cancer – A number of genes have been implicated in kidney cancers that are sporadic (not inherited) and/or that develop as a result of hereditary mutations (Linehan, 2012). The more significant genes are listed below (From: Linehan, 2012):
- VHL – the predominant cancer gene in sporadic clear cell kidney cancers. A mutation in VHL leads to an increase in the hypoxia-inducible transcription factors HIF1A and HIF2A (also known as EPAS1) that leads to increased transcription of many downstream genes that include KDR (VEGFR), PDGFRB, EGFR, and SLC2A1. VHL +/+ clear cell kidney cancer is caused by different gene(s) than VHL -/- clear cell kidney cancer. Normal VHL is involved in cellular oxygen sensing. Mutations are found in both hereditary and sporadic cases.
- PBRM1 – involved in sporadic clear cell kidney cancer. Truncating mutations of PBRM1, the SWI/SNF chromatin remodeling complex gene, in one study were found in 41% of tumors.
- FH – involved in HLRCC papillary kidney cancers, and codes for the enzyme fumarase (fumarate hydratase) that is involved in the TCA cycle.
- FLCN – Mutations in the FLCN gene are associated with BHD but not sporadic chromophobe kidney cancers and oncocytomas. It is involved in the LKB1/AMPK and mTOR pathways. FLCN encodes the protein folliculin.
- MET – mutations in the MET gene have been associated with some sporadic papillary kidney cancers (about 13%).
- BAP1 – found to be mutated in about 15% of ccRCC and it is largely mutually exclusive with PBRM1 mutations. Both also have distinct gene expression signatures. Tumors with the BAP1 mutation have been correlated with aggressive pathogenicity that includes high Fuhrman grade and tumor necrosis.
- SDHB – germline mutations in SDHB have been found in a minority of RCC patients. (Ricketts et al., 2008).
In a survey of more than 400 ccRCC tumors, 19 significantly mutated genes were identified. The eight most extreme were those of VHL, PBRM1, SETD2, KDM5C, PTEN, BAP1, MTOR, and TP53. SETD2 is involved in histone modification as a methyltransferase. (The Cancer Genome Atlas Research Network, 2012). PBRM1 encodes a protein subunit of the PBAF SWI/SNF chromatin remodeling complex (Wang et al., 2014).
Some kidney cancers can be characterized by mutations in the MiT transcription factors family of genes: TFE3, TFEB, and MITF, and these tumors possess a distinct histology as well. Germline mutations of MITF have been discovered in the germline of patients with either melanoma or kidney cancer or both. TFE3 and TFEB are involved in translocations of certain chromosomes that are primarily seen in children and young adults (chromosome 1 and chromosome X with TFE3 and chromosome 6 and chromosome 11 with TFEB) (Yang et al., 2014).
Chromosomal translocations and other aberrations: Certain chromosomal differences have been identified with some of the subtypes of kidney cancer. They are:
- ccRCC: Identified by the specific deletion of chromosome 3p, as well as a VHL mutation. Duplication of the chromosome band 5q22 and deletion of chromosomes 6q, 8p, 9p, and 14q also occur.
- Papillary RCC: Associated with trisomies of chromosomes 3q, 7, 8, 12, 16, 17, and loss of the y chromosome.
- Chromophobe RCC: Has a combination of loss of chromosomes 1, 2, 6, 10, 13, and 17.
From: Ljungberg et al., 2013.
Classification of Kidney Cancer
Histological classification of Kidney Cancer
Renal cell carcinoma (RCC): The most common type of kidney cancer is renal cell carcinoma (RCC), which develops in the lining of the tubules and accounts for about 90% of all cases. There are several subtypes of RCC based on histopathological classification of the tumor by section under microscope. They include the following.
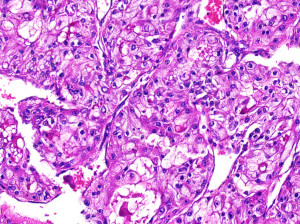
Note that the cells in this kidney cancer specimen have clear cytoplasm, hence a common name of this tumor is clear cell cancer
- Clear cell (conventional) RCC: The most common form, it represents roughly 70% of all cases. Tumor cells look very pale or clear under the microscope. Clear cell RCC is associated with the von Hippel Lindau (VHL) gene mutation that is seen in hereditary kidney cancer, and 70% of all non-hereditary cases have a VHL mutation as well. Prognosis is good if the tumor has not spread (Kidney Cancer Center, 2013).
- Papillary (chromophil) RCC: The second most common form, it represents 10-15% of all cases. Some of the tumors form small finger-like projections (papillae), and cells stain pink. There are two subtypes based on the appearance of the cells: Type I (5%) and Type II (10%). Hereditary forms of both exist, and there is an increased incidence among African Americans and of bilateral disease (affecting both kidneys) associated with the subtype as a whole. Prognosis is good if the tumor has not spread (Kidney Cancer Center, 2013).
- Chromophobe RCC: The third most common form, but rare, it accounts for about 5% of all cases. Cells are pale but much larger than those of Clear Cell RCC and they possess other distinctive features as well. It is thought to originate from the same cell type as that which forms renal oncocytomas (a benign form of kidney cancer), and hybrid tumors that possess features of both have been diagnosed. There is also an inherited form of chromophobe RCC in association with renal oncocytoma known as Birt Hogg Dubé syndrome that is associated with FLCN, a tumor-suppressor gene. It rarely metastasizes, and the prognosis is good (Kidney Cancer Center, 2013).
- Collecting Duct Carcinoma: Rare but aggressive, it accounts for less than 1% of all cases. The cancer cells of this subtype can form irregular tubes. It is more common in younger individuals and is usually metastatic when diagnosed. Chemotherapy is usually the method of treatment as this subtype does not respond to conventional therapies for RCC (American Cancer Society 2011).
- Unclassified RCC: Unclassified forms of RCC account for as much as 5% of all cases. They have genetic and structural features that do not match those of the other subtypes, and they tend to be aggressive (American Cancer Society 2011).
- Medullary RCC: Also rare and aggressive, it is thought to be a variant of collecting duct carcinoma, and is commonly associated with the sickle cell trait, thus making it more common among African Americans. Chemotherapy is the primary method of treatment.
- Sarcomatoid RCC: This can occur with any of the RCC subtypes and is known as “differentiation.” It is diagnosed when some of the RCC cells have the appearance of sarcoma cells. It is frequently found in patients whose kidney cancer has widely metastasized. The prognosis is poor and chemotherapy is only sometimes used to treat it.
- Other rare forms: These include, but are not limited to, multilocular cystic RCC, mucinous tubular and spindle cell carcinoma, translocation RCC, and neuroblastoma-associated RCC, and each causes less than 1% of RCCs.
Other Kidney Cancers:
- Transitional cell carcinoma (TCC): About 5-10% of kidney cancer cases are transitional cell carcinomas, also known as urothelial carcinomas. These develop in the transitional cell lining of the renal pelvis, and the cells look much like bladder cancer cells and the cancer acts much like bladder cancer, but the signs and symptoms are typically the same as those of other kidney cancers. As with bladder cancer, however, TCC is often linked to cigarette smoking and occupational exposure to certain carcinogenic chemicals. Prognosis is poor if the tumor is large or has metastasized.
- Wilms Tumor (nephroblastoma): A rare form of cancer with only about 500 new cases diagnosed every year in the United States. It usually occurs in children and is rarely found in adults (American Cancer Society 2011).
- Renal Sarcoma: This cancer comprises less than 1% of all cases of kidney cancer, and they arise in the blood vessels or connective tissue of the kidney (Kidney Cancer Center, 2013).
Grading:
Kidney cancers are given a Fuhrman grading of 1-4 based on the resemblance of the cancer cell nuclei to the nuclei of normal kidney cells. Grade 1 cancer cell nuclei still closely resemble those of normal kidney cell nuclei, and the cancer prognosis is good. Grade 4 look much different and the prognosis is worse.
Classification based on stage:
TNM staging is used for kidney cancer. It is based on the size of the tumor (T), the number of lymph nodes affected (N), and the extent of metastasis of the cancer (M) (Kidney Cancer Center, 2013).
Classification of kidney cancer based on gene expression:
A recent study has discerned four subtypes of ccRCC, detected with mRNA and miRNA expression data sets. Distinct gene groups defined as over-expressed, under-expressed, weakly over-expressed, and weakly under-expressed led to the identification of the four subtypes. A strong association between the up-regulated genes and cellular responses to tumor development was detected. The down-regulated genes were implicated in compromises of kidney function from the cancer. Pathway and network analyses that were also conducted provided supporting evidence for four subtypes of ccRCC. Among the significant networks that were identified, NF-κB and UBC were found to be central nodes for molecular transport, metabolic disease network, and renal or urological disease network, as well as for hereditary disorder. The expression levels of both were not significantly different in ccRCC compared to normal levels, but due to the extent with which they interconnect with other genes in the network, they seem to have important regulatory roles in the cancer (Escudier et al., 2010).
Another study identified two distinct subtypes of ccRCC based on consensus cluster analysis (Brannon et al. 2010). The subtypes, designated clear cell type A (ccA) and type B (ccB), were found to have a similar frequency of VHL inactivation but activation of different dominant biological pathways that results in different patterns of gene expression. Type A tumors had significantly improved disease-specific survival compared to type B, and type A was more often associated with grade 1 and stage 1 tumors whereas type B was more often associated with grade 4 and stage 4 cancers (Yang et al., 2014).
Classification based on metabolic pathways
Different RCC subtypes also are associated with different metabolic pathways which can contain potential targets for targeted therapies (Escudier et al., 2010):
- The hypoxia-inducible pathway: clear cell and papillary type II with fumarase
- The mTOR signaling pathway: clear cell and papillary type II
- The c Met-RAF-MEK-ERK pathway: papillary type I and translocation RCC (a rare subtype)
- The c-kit-RAF-MEK-ERK pathway: chromophobe RCC
Diagnosis and Treatment
An ultrasound, CT scan, and MRI, among other tests, can all be ordered to detect kidney cancer or metastasis. Stages I, II, and III tumors are usually surgically removed, although tumor ablation (destruction of the tumor without removal of it) is sometimes used instead. Stage IV, in which cancer has spread farther than to regional lymph nodes, may be treated with surgical excision of the tumor(s), but because the cancer has spread to other organ(s), treatment options most often include immunotherapy and targeted treatments, and sometimes radiation therapy and chemotherapy.
Immunotherapy
IL-2 and interferon: Immunotherapy, also called biologic therapy, is the utilization of the patient’s immune system to attack the cancer. Interleukin-2 (IL-2, brand name Proleukin®) and interferon-α (IFN-α) have been the most commonly used immunotherapy agents (Escudier, 2012). Approximately 20% of patients have some degree of tumor regression with immunotherapy, and about 5-7% of patients have complete tumor regression usually due to high dose IL-2 protocol; however, due to toxicity concerns, high-dose IL-2 should be considered only for young and fit patients who can tolerate the therapy (Ibid.). Use of IL-2 is also called cytokine therapy. In addition, IL-2 has not been shown to significantly improve survival in patients who have RCC. Interferon-α seems to be less effective when used alone, leading to its more common usage in combination treatment with the targeted drug bevacizumab (Avastin®) (Ibid.).
Drugs that block PD-L1 and PD-1: An experimental approach is the use of drugs that block the PD-L1 protein on cancer cells, which helps them evade the immune system, and the corresponding PD-1 protein on T cells of the immune system. In early studies, an anti-PD-1 drug called nivolumab (BMS-936558) shrank tumors in about 25% of patients with RCC, and another drug (BMS-936559) that targeted PD-L1 shrank tumors in about 10% of patients (Escudier, 2012).
Experimental vaccines: One approach is to remove immune system cells, treat them with cytokines and expose them to dead tumor cells to make active dendritic cells, following which the dendritic cells are injected into the patient to stimulate the immune system to respond to the cancer. This vaccine is known as AGS-003, and it has been associated with improved survival when used in combination with the targeted drug sunitinib compared to use of sunitinib alone. The vaccine IMA901, developed from 10 naturally presented tumor-associated peptides, has been found in a phase II clinical study to improve survival rates. MVA5T4, which utilized a modified vaccinia virus to induce an immune response to cells that express the 5T4 antigen (found in most tumors), was not found to be effective (Escudier, 2012).
T-cell modulation: Anti PD-L1 and PD-1 drugs (see above) are examples of T-cell modulation. Other examples include the use of anti-CTLA-4 antibody, denileukin difitox, and soluble LAG-3 (Escudier, 2012).
Targeted Therapies
All the targeted therapies for kidney cancer are anti-angiogenic. Kidney cancer is highly angiogenic and vascularized. Tumors secrete vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF) that activate tyrosine kinases, proteins that are involved in the development of blood vessels that enable tumors to grow and metastasize (He et al., 2014). Some agents target tyrosine kinases (multikinase inhibitors) and some target mTOR, a protein that is also involved in angiogenesis (National Cancer Institute, 2004).
Drugs that block tyrosine kinases (National Cancer Institute, 2004):
- Axitinib (Inlyta®) – a highly selective, potent inhibitor of VEGF receptors 1, 2, and 3 (VEGFR-1, VEGFR-2, and VEGFR-3); is administered orally
- Pazopanib (Votrient®) – blocks VEGFR-1, VEGFR-2, VEGFR-3, PDGFR, and c-KIT and is administered orally
- Sorafenib (Nexavar®) – blocks cRAF, bRAF, KIT, FLT-3, VEGFR-2, VEGFR-3, and PDGFR-β and is administered orally
- Sunitinib (Sutent®) – blocks VEGFR-1, VEGFR-2, PDGFR, c-Kit and is administered orally
Drugs that block mTOR (National Cancer Institute, 2004):
- Everolimus (Afinitor®) – blocks mTOR and is used after other drugs such as sorafenib and sunitinib have been tried; administered orally
- Temsirolimus (Torisel®) – blocks mTOR and is administered intravenously
A drug that binds VEGF (National Cancer Institute, 2004):
- Bevacizumab (Avastin®) – a biologic antibody that binds directly to VEGF to inhibit angiogenesis; administered intravenously; may be used in combination with interferon-α
Considerations for Researchers
Kidney Cancer Sample Availability
While the majority of kidney cancer cases are detected early, biopsies are not usually performed, as imaging tests typically provide the physician with enough information to decide if surgery should be performed. Historically, the availability of kidney cancer samples and tissue for early stages should be good because early stage kidney cancers are often surgically removed. However, if you are searching for kidney cancer samples collected prior to treatment, please note that recent trends have moved towards treatment prior to surgery, so your chances of finding untreated samples may be better if you are working with an organization that has access to CAP graduated archival samples. Surgical resection is often performed for metastatic renal cell carcinoma as well, so tissue samples may be available, excepting in the case of sarcomatoid RCC as patients with this type do not usually benefit from surgery (American Cancer Society 2011; Thompson, 2009). Biopsies for stage III and IV may be available but are hard to obtain because they are often used by the clinic and may not ever reach the clinic’s repository. Blood products may be available on a prospective basis.
For kidney cancer samples collected prospectively, plan ahead as collection times can exceed several months depending on your inclusion and exclusion criteria. Biomarker information is only available if it was collected as part of standard care at the time of treatment. Data mining fees may apply if you require specific biomarker criteria. Remember that many biorepositories and biobanks contain primarily FFPE samples that have been CAP graduated, which means that they are at ten years old, and data associated with the samples will reflect standard care as it was a decade ago.
The frequency of blood draws from cancer patients makes it relatively easy to prospectively collect blood derived kidney cancer samples like plasma and serum, but scientists need to be realistic about requesting volumes greater than 1 ml, custom collection protocols, or anything else that deviates from standard care. The more your collection requirements deviate from standard care, and the the harder the request will be to fill.
Biomarker screening of samples is possible, but this is unlikely to be cost effective unless you want to purchase all tested specimens regardless of their biomarker profile. If you only want kidney cancer samples with certain markers, remember that you will have to pay for testing of the ones that are negative too. A better alternative may be to purchase sections only for a suitable number of cases, screen them in your own lab using your preferred assay or third party, high volume genetic screening service, then request the blocks of interest.
References
American Cancer Society. (2011). Kidney Cancer. ACS. Retrieved from:http://www.cancer.org/cancer/kidneycancer/.
Brannon, A. R., Reddy, A., Seiler, M., Arreola, A., Moore, D. T., Pruthi, R. S., … Rathmell, W. K. (2010).
Molecular Stratification of Clear Cell Renal Cell Carcinoma by Consensus Clustering Reveals Distinct Subtypes and Survival Patterns. Genes & Cancer, 1(2), 152–163. doi:10.1177/1947601909359929
The Cancer Genome Atlas Research Network. (2013). Comprehensive molecular characterization of clear
cell renal cell carcinoma. Nature, 499(7456), 43-49. doi:10.1038/nature12222
Urology Health. 2011. Kidney Cancer. Urology Health. Retrieved from:
http://www.urologyhealth.org/urology/index.cfm?article=24
Escudier, B. (January 01, 2012). Emerging immunotherapies for renal cell carcinoma. Annals of Oncology:
Official Journal of the European Society for Medical Oncology / Esmo, 23, 35-40.
Escudier, B., Kataja, V., & ESMO Guidelines Working Group. (January 01, 2010). Renal cell carcinoma:
ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology : Official Journal of the European Society for Medical Oncology / Esmo, 21, 137-9.
He, D., Li, L., Zhu, G., Liang, L., Guan, Z., Chang, L., Chen, Y., … Chang, C. (August 13, 2014). ASC-J9
Suppresses Renal Cell Carcinoma Progression by Targeting an Androgen Receptor-Dependent HIF2 /VEGF Signaling Pathway. Cancer Research, 74, 16, 4420-4430.
Kidney Cancer Association (2013). “About Kidney Cancer.” KCA. Retrieved from: http://www.kidneycancer.org/knowledge/learn/about-kidney-cancer/
Linehan, W. M. (January 01, 2012). Genetic basis of kidney cancer: role of genomics for the development
of disease-based therapeutics. Genome Research, 22, 11, 2089-100.
Ljungberg, B. et al. (March 2013). Guidelines on Renal Cell Carcinoma. European Association of Urology.
Retrieved from: http://uroweb.org/wp-content/uploads/10-Renal-Cell-Carcinoma_LR.pdf
National Cancer Institute. (2004). Kidney Cancer. NCI at the National Institute of Health. Retrieved from:
http://www.cancer.gov/cancertopics/types/kidney
Ricketts, C., Woodward, E. R., Killick, P., Morris, M. R., Astuti, D., Latif, F., & Maher, E. R. (January 01,
2008). Germline SDHB mutations and familial renal cell carcinoma. Journal of the National Cancer Institute, 100, 17, 1260-2.
SEER. 2005. SEER Stat Fact Sheets: Kidney and Renal Pelvis Cancer. National Cancer Institute:
Surveillance, Epidemiology, and End Results Program. Retrieved from: http://seer.cancer.gov/statfacts/html/kidrp.html.
Thompson, J. A. (January 01, 2009). Metastatic renal cell carcinoma: current standards of care. Clinical Journal of Oncology Nursing, 13, 8-12.
Wang, M. H., Huang, C.H., Zheng, T., Lo, S.H., and Hu, I. 2014. Discovering pure gene-environment
interactions in blood pressure genome-wide association studies data: a two-step approach incorporating new statistics. Biomed Central Proceedings. 8(Suppl 1): 562 doi:10.1186/1753-6561-8-S1S62. Retrieved from: http://www.genecards.org/cgi-bin/carddisp.pl?gene=PBRM1.
Wiley. (2014). Low cholesterol linked with worse survival in patients with kidney cancer. ScienceDaily.
Retrieved from: www.sciencedaily.com/releases/2014/06/140612085759.htm.
Yang, J. Y., Dunker, A. K., Liu, J. S., Qin, X., Arabnia, H. R., Yang, W., … & Yang, M. Q. (2014). Advances in translational bioinformatics facilitate revealing the landscape of complex disease mechanisms. BMC Bioinformatics, 15(Suppl 17), I1.